 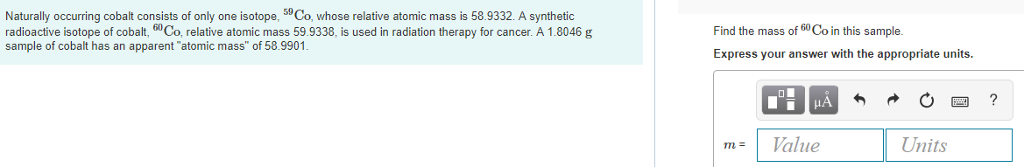
This is why the decay from the $J=4$ state to the $J=0$ ground state can only be done through two separate gamma-decay events, instead of a direct decay to the ground state. Beta decay - is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming th. However, as with beta decay, there is an upper limit to how much angular momentum each gamma decay can carry away. Most Co-60 that is ingested is excreted in the feces however, a small amount is absorbed by the liver, kidneys, and bones. It is also possible for the beta decay to remove a whole three units, leaving the nickel daughter nucleus at a lower excited state with $J=2$ and less energy, but this is hard to achieve and does not happen very often.įinally, most of the time the nickel-60 decay product still needs to get rid of its energy and angular momentum, and it can only do that via gamma decay. Instead, as captured in the schematic, the most likely process is that the beta decay will only be able to reduce the angular momentum quantum number by one, leaving an excited nickel-60 nucleus with $J=4$. These can combine in different ways to produce different total angular momentum removed from the system, but there is a limit to this $-$ and it is extremely hard to remove five units of angular momentum at once. the relative (orbital) angular momentum of the electron, neutrino, and daughter nucleus.The initial beta decay can get rid of angular momentum in several different ways: (The angular momentum quantum number is indicated to the right of each line, always followed by the parity, $+$ or $-$, of the state.) The decay schematic in Wikipedia explains how this process pans out: This means that the decay process needs to get rid of (1) a bunch of excess energy, and also, more importantly, (2) a lot of angular momentum. The reason for this double emission is that cobalt-60 has an angular momentum quantum number $J=5$, while the ground state of nickel-60 has angular momentum quantum number $J=0$. This nickel-60 decay product is then left in an excited state, and it is this excited state that produces two separate gamma-ray emissions. The alpha particle removes two protons (green) and two neutrons (gray) from the uranium-238 nucleus. For both isotopes, the gamma rays energies are. 60 Co produces gamma rays with energies of 1.173 and 1.332 MeV and has a half-life of 5.27 years, whereas 137 Cs produces gamma rays with an energy of 0.662 MeV and has a longer half-life of 30.1 years. These sorts of cascades are the bread and butter of nuclear physics the double-photon decay paper you found is much rarer.Cobalt-60 does not directly decay by gamma decay $-$ instead, it decays by beta decay to nickel-60. Figure 1: A nucleus of uranium-238 (the parent nuclide) undergoes decay to form thorium-234 (the daughter nuclide). Cobalt-60 (60Co) and caesium-137 ( 137 Cs) are the most widely used sources of gamma radiation. That photon must carry lots of orbital angular momentum, in addition to its spin, so the first excited state of cobalt is a relatively long-lived isomer (about ten minutes).Ĭobalt-60 is used as a gamma source because it decays to an excited state of the nickel-60 nucleus, which then cools by emitting a sequence, or a "cascade," of photons. Sum of superscripts : 211 y + 0, so y 211. The equation for the beta decay of 60Co is: 2760Co -> 2860Ni. The main point to remember in balancing nuclear equations is that the sums of the superscripts and the subscripts must be the same on each side of the equation. This problem has been solved You'll get a detailed solution from a subject matter expert that helps you learn core concepts. What is the nuclear decay equation for cobalt-60 Wiki User. Complete the nuclear equation in your answer book. Explain in terms of BOTH protons and neutrons, why Co-59 and Co-60 are isotopes of cobalt. Click in the answer box to activate the palette. The incomplete equation for the decay of cobalt-60, including beta and gamma emissions, is shown below. Since cobalt-60 has ground state spin-parity $5^+$, first excited state $2^+$, a single photon can mediate the transition. Use nuclear symbols to write a nuclear equation to represent cobalt-60 decaying to nickel-60, plus a beta particle, plus a gamma ray. 
Usually, in nuclear decays, magnetic-dipole transitions are suppressed compared to electric-dipole transitions. The paper you have linked measures a rare mode where two real photons are produced, and a surprising observation that in the double decay $E$-type photons are produced at the same rate as $M$-type photons. Mostly they decay by emitting a "virtual" photon, which produces a real positron-electron pair in the field of the nucleus. Since a single photon must carry away at least one unit of spin, these excitations cannot decay by one-photon emission. Both of these nuclides have a first excited state with spin-parity $0^+$, the same as their ground state. The paper that you cite describes decays in calcium-40 and zirconium-90 by emission of two photons at once. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
